It’s time to give patients with pleural effusions a higher standard of care.
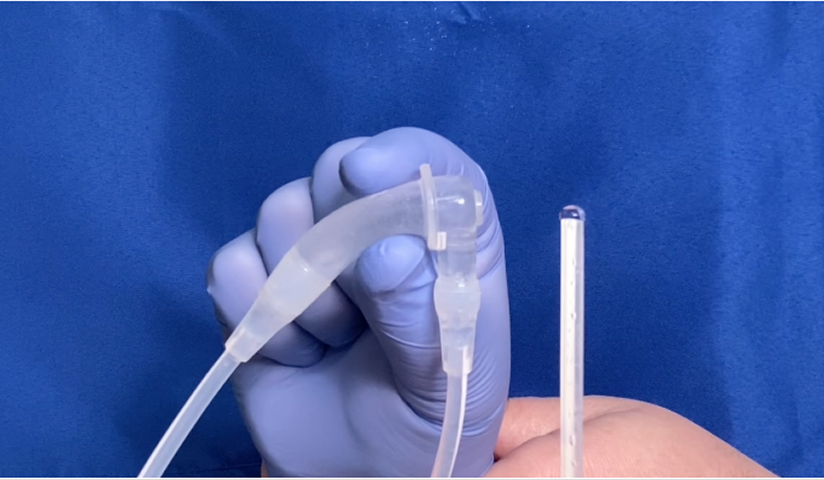
How ACES™ works
Just 0.5 mm of compression moves a drop of fluid: meaning roughly 23,040 breaths a day can move up to 1.2 liters of fluid per day
Natural breathing motion gives patients the freedom to breathe life in
ACES™ (Automatic Continuous Effusion Shunt) uses respiratory motion to drive continuous, automatic transfer of pleural effusion fluid into the abdomen where it is naturally reabsorbed—without the need for pleurodesis or external drainage catheters.1.2

10
Average IPC at-home drainage procedures per month4
VS

0
ACES at-home drainage procedures per month3
A single implant, performed inpatient or outpatient1,2
The implant is placed in a straightforward procedure with an expected hospital stay of one day or less.
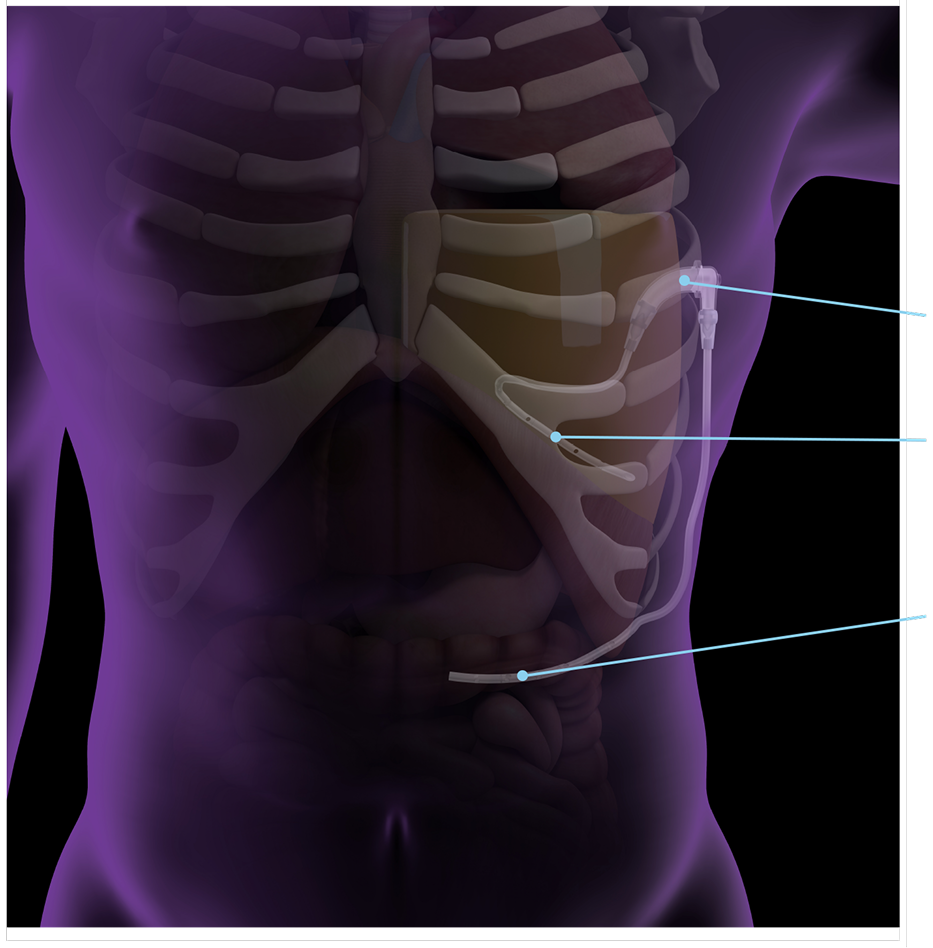
Pump Chamber
Pleural Catheter
Peritoneal Catheter
Minimally invasive
Through a small incision, the pump chamber is placed under the skin between the ribs. One catheter is positioned in the pleural space, the other in the abdomen for drainage.
Continuous relief
The pump continuously shunts fluid from the pleural space to the abdomen—without the need for external catheters.
Easy to adopt, with a familiar technique2
Drawing on two familiar procedures—chest tube placement and peritoneal dialysis catheter insertion—the ACES implant technique is designed to feel intuitive with minimal training. Clinicians routinely report the procedure is “easy to perform” and fits naturally into current practices.
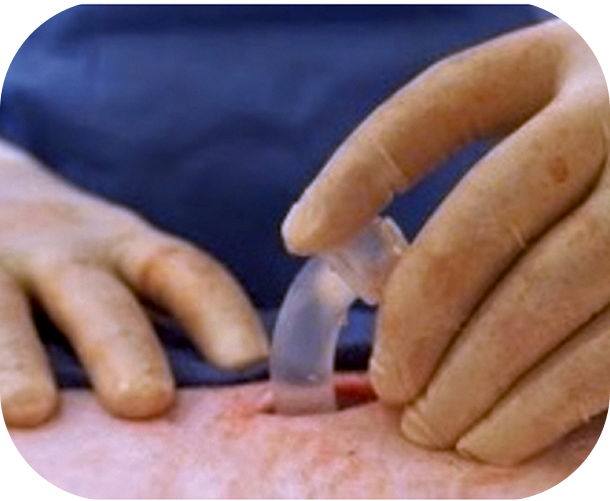
Pleural entry
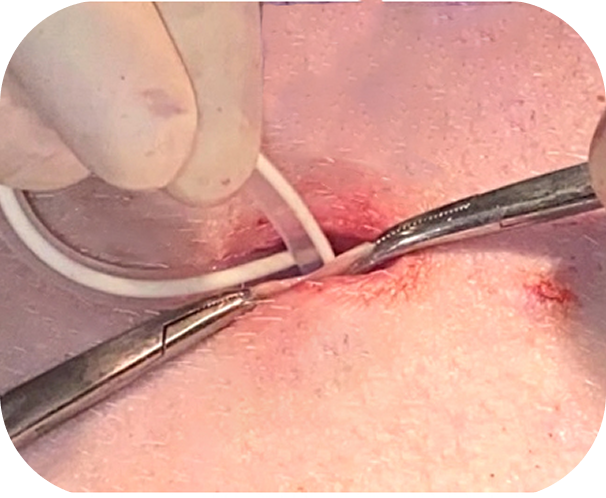
Peritoneal entry
Low maintenance, with no external hardware, tubes or bottles1,2
ACES eliminates the need for at-home fluid management and helps lower the overall cost of pleural effusion care.

“Providing our patients with a solution that improves their quality of life and lessens the burden of maintaining an implanted medical device for their chronic medical condition has the potential to really change the way we manage pleural disease.”
—Dr. Jason Akulian, MD, Chapel Hill, NC
Consider ACES for these patients

Favorable patient criteria:
- Simple effusions
- Life expectancy >3 months
- They are or can be mobile

Patients who have received ACES:
- Lung cancer patients
- Breast cancer patients
- Renal failure patients
- Patients with idiopathic pleural effusions
Patients who have received ACES:
ACES helps reduce the need for intensive IPC oversight—offering a level of prevention that may lower infection risk compared to traditional IPC management.
Pre-op considerations
- General anesthesia risk should be factored
- Previous abdominal surgeries should be well understood
- Expandable (not trapped) lungs may have better effusion drainage
- Ascites may limit resorption
- Sarcoma or mesothelioma may spread along catheter tracts
Explore the latest clinical evidence supporting ACES therapy
References: 1. Data on File. Pleural Dynamics. The ACES Study for Aseptic Pleural Effusion. ClinicalTrials.gov ID NCT06210685. 2. Automatic and Continuous Effusion Shunt (ACES) Instructions for Use. P/N 01127, Rev. A. Release 2025. 3. Thiboutot J, Block MI, Akulian C, et al. A prospective study evaluating fluid shunting using the automatic continuous effusion management system in patients with symptomatic recurrent pleural effusion: The ACES Study. Chest. 2025;168(4):A7305-A73065 4. Muruganandan S, Azzopardi M, Fitzgerald DB, et al. Aggressive versus symptom-guided drainage of malignant pleural effusion via indwelling pleural catheters (AMPLE-2): an open-label randomised trial. Lancet Respir Med. 2018;6(9):671-680.
The ACES implant is approved for sale in the USA. Caution: Federal (United States) law restricts this device to sale by or on order of a physician. Prior to use, please see the Instructions for Use for a complete listing of Indications, Contraindications, Warnings, Precautions, Potential Complications, and Insertion Instructions.

